
As of early 2026, more than 287 drugs are in short supply across the United States - nearly half of them critical medications for heart attacks, seizures, cancer treatments, and infections. Hospitals are rationing painkillers. Pharmacies are calling patients to switch to less effective alternatives. And doctors are making life-or-death decisions because the medicine they need simply isn’t there. This isn’t a temporary glitch. It’s a system failure. And Congress is finally trying to fix it - with two bills that could change everything, if they ever get out of committee.
The Two Bills Trying to Stop Drug Shortages
The most direct response so far is the Drug Shortage Prevention Act of 2025 (S.2665). Introduced by Senator Amy Klobuchar in August 2025, this bill doesn’t promise new money or big reforms. Instead, it asks one simple thing: manufacturers must tell the FDA when demand for a critical drug starts to spike. Right now, companies don’t have to report anything until the shortage is already happening - often too late. This law would force them to give early warnings, giving regulators time to step in before patients go without.
It sounds basic. But it’s not. Right now, manufacturers can sit on production delays, ignore supply chain warnings, or wait until a drug runs out before saying anything. The FDA’s Drug Shortage Portal, which tracks these issues, is already overwhelmed. In October 2025, 63% of shortages were traced back to manufacturing delays - not raw material shortages or regulatory delays. If companies had to report early signs of trouble, the FDA could push for backup suppliers, adjust distribution, or even fast-track approvals for alternatives. But that only works if the agency is running.
And that’s the problem.
The Shutdown That Broke Everything
Since October 1, 2025, the U.S. government has been shut down - the longest in history. Nearly 800,000 federal workers are furloughed. The FDA’s drug shortage team? Not working. Their computers? Not maintained. The portal that tracks shortages? Glitching. The people who review emergency requests for new suppliers? On unpaid leave.
Even if S.2665 passed tomorrow, it couldn’t be enforced. The agency that’s supposed to make it work doesn’t have staff. No one is answering phones. No one is checking emails. No one is updating the list of which drugs are running out. And without that, the bill is just words on paper.
Meanwhile, the second bill - the Health Care Provider Shortage Minimization Act of 2025 (H.R.1160) - is even more mysterious. All we know is the title. No sponsor list. No committee assignment. No text published anywhere. It’s supposed to address the shortage of doctors, nurses, and primary care providers, which affects 122 million Americans living in areas with no nearby clinic. But without knowing what’s in it, we can’t say if it would help. Will it fund more medical schools? Expand telehealth? Pay doctors to work in rural areas? No one knows. And with Congress frozen, it’s not even being discussed.
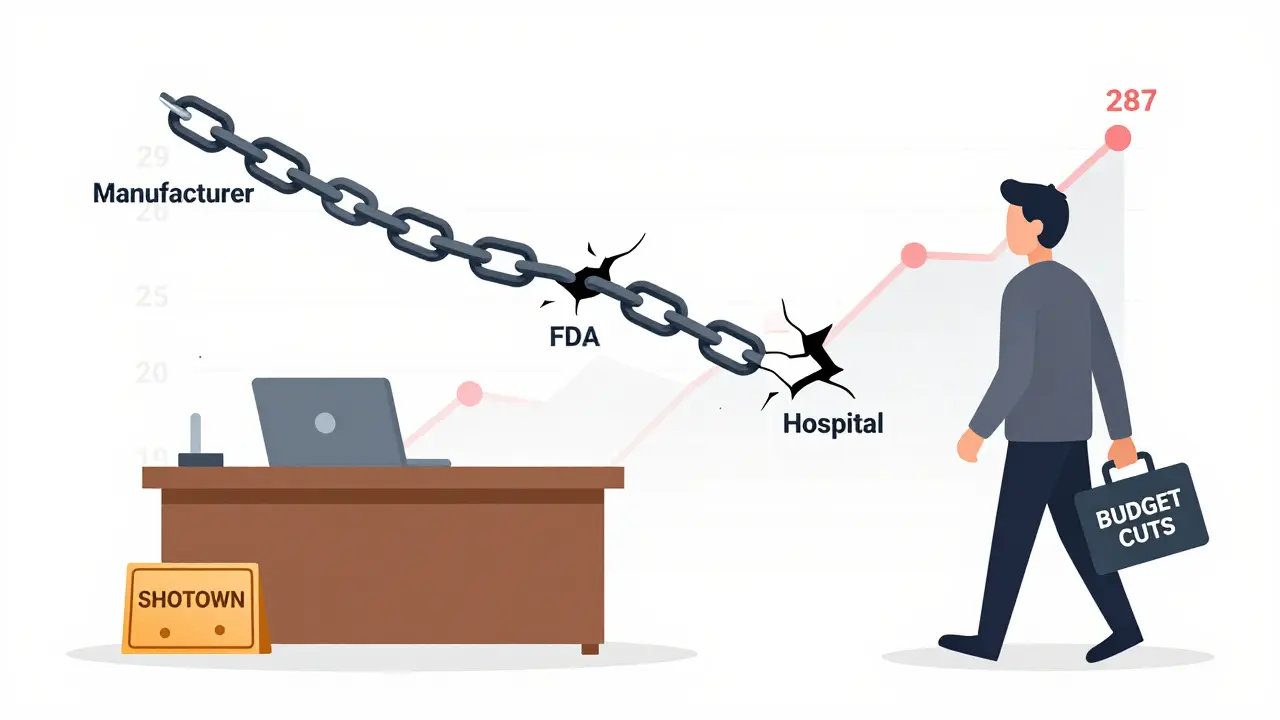
Why This Keeps Happening
Drug shortages aren’t new. But they’ve gotten worse because the system is built to ignore the problem until it’s an emergency. Manufacturers rely on thin margins, especially for generic drugs. If a drug costs $1 a pill and you make 10 million pills, your profit is $10 million - after paying for ingredients, labor, and compliance. That’s not much. So when a factory has a minor equipment issue, or a supplier raises prices, it’s cheaper to wait and let the shortage happen than to fix it early.
Then there’s the lack of backup. The U.S. depends on just a few global suppliers for key ingredients. One plant in India or China shuts down - and suddenly, half the country’s supply of antibiotics disappears. The FDA knows this. But they don’t have the power to force companies to diversify suppliers. And without that, shortages keep coming.
And it’s not just drugs. Hospitals reported in October 2025 that 98% had at least one critical drug shortage in the last quarter. One hospital in Ohio had to delay cancer treatments because the chemo drug they needed was out. Another in Texas switched patients to a less effective painkiller - and saw more emergency room visits because it didn’t work as well.
What’s Being Done - and What’s Not
Meanwhile, Congress is busy with other things. In November 2025, House Republicans pushed to reverse a rule that lets senators sue over phone records. The Stop Secret Spending Act got attention - but it’s about Treasury transparency, not medicine. The Rescissions Act of 2025 cut $7.9 billion from foreign aid. But not one cent was added to fix drug shortages.
The Congressional Budget Office says implementing S.2665 would cost $45 million a year - a fraction of what’s being cut elsewhere. But with the federal deficit hitting $1.74 trillion in the last fiscal year, no one wants to spend money on a problem that doesn’t make headlines. And right now, the shutdown is the headline. The drug shortages? They’re background noise.
Doctors know this. A September 2025 survey by the American Medical Association found that 87% of physicians have seen patients suffer because of drug shortages. But only 12% even knew H.R.1160 existed. The public? Most have no idea. They just notice their prescription is gone - and their doctor says, "I’m sorry, we’re out."

What Needs to Happen Next
If these bills are going to work, three things need to change:
- Get the government open. No law can be enforced if the FDA is shuttered. The shutdown must end before any of this moves forward.
- Make the bill real. S.2665 needs clear definitions: What counts as a "critical drug"? What’s the deadline for reporting? What penalties apply if companies lie or delay? Right now, it’s all vague.
- Build real backup systems. The U.S. can’t keep relying on one factory in one country. The government needs to incentivize domestic production of key ingredients - not just wait for shortages to happen.
There’s no magic fix. But without early warnings, without enforcement, and without political will, shortages will keep getting worse. And patients will keep paying the price.
What You Can Do Right Now
If you’re on a medication that’s been in short supply, don’t wait for Congress to act. Talk to your pharmacist. Ask if there’s a therapeutic alternative. Check the FDA’s Drug Shortage website - it’s still updating, even during the shutdown. Join patient advocacy groups. Call your representative. Tell them you’ve been affected. The only reason these bills get attention is because people demand it.
Because right now, the system is broken. And unless enough people push back, it won’t get fixed - not this year, not next year, and maybe not ever.
What is the Drug Shortage Prevention Act of 2025?
The Drug Shortage Prevention Act of 2025 (S.2665) is a proposed law that would require pharmaceutical manufacturers to notify the FDA when demand for critical drugs increases. The goal is to give regulators early warning so they can prevent shortages before they happen. The bill is currently in the Senate Health, Education, Labor, and Pensions Committee and has not yet been voted on.
Why hasn’t the bill passed yet?
The bill is stuck because the U.S. government has been shut down since October 1, 2025 - the longest shutdown in history. Federal agencies like the FDA are not operating normally, so even if the bill passed, there would be no one to enforce it. Additionally, Congress is focused on budget battles and partisan issues, leaving public health legislation sidelined.
How many drugs are currently in short supply?
As of September 30, 2025, the FDA listed 287 drugs in shortage, with 47% classified as critical - meaning they’re used to treat life-threatening conditions like cancer, heart failure, or severe infections. The number has been rising steadily since 2020.
What’s the Health Care Provider Shortage Minimization Act?
H.R.1160 is a bill introduced in the House to address the shortage of doctors, nurses, and other healthcare workers. But as of early 2026, no detailed text, sponsors, or committee assignments have been made public. Without knowing what’s in it, it’s impossible to assess its potential impact.
Can I find out if my medication is on the shortage list?
Yes. The FDA maintains a public Drug Shortages list on its website (FDA.gov). Even during the government shutdown, the site continues to update with new shortages reported by manufacturers and healthcare providers. You can search by drug name or browse by category.



pradnya paramita
February 5, 2026 AT 02:53The Drug Shortage Prevention Act of 2025 (S.2665) represents a structural intervention at the supply-side information asymmetry level. Mandating pre-emptive notification thresholds for critical drug demand surges shifts the paradigm from reactive triage to anticipatory supply-chain orchestration. The FDA’s current operational latency-particularly during the shutdown-exacerbates systemic fragility. Without real-time telemetry from manufacturers, regulatory intervention is inherently lagging. We need blockchain-anchored production dashboards, not just Excel spreadsheets.
Furthermore, the lack of tiered criticality definitions undermines enforcement. Is insulin classified the same as vecuronium? No. Yet the bill doesn’t differentiate. This is regulatory sloppiness masquerading as policy. Define criticality by ATC code, therapeutic index, and patient volume thresholds-not vague moral appeals.
Wendy Lamb
February 5, 2026 AT 10:52My mom’s on chemo. She had to switch to a less effective drug last month. Her oncologist said it’s not just about availability-it’s about how the body reacts. We need better alternatives, not just more notifications.
Antwonette Robinson
February 5, 2026 AT 12:50Oh wow, so now we’re going to make drug companies into federal snitches? Next they’ll require them to file weekly reports on how many people they didn’t kill this week. Meanwhile, the FDA’s been on vacation since October. Did anyone tell the bill it’s 2026 and not 2012?
Ed Mackey
February 5, 2026 AT 15:59I work in a rural clinic. We’ve been out of epinephrine for 3 weeks. No one calls us back. The FDA site says ‘under review’ but no one’s reviewing anything. I don’t care about bills-I care about someone answering the phone. This shutdown is killing people and no one’s talking about it like it’s a real thing.
Alex LaVey
February 6, 2026 AT 04:09Let’s not forget the human side here. I’ve talked to nurses in Texas who are crying because they have to tell a 12-year-old with leukemia, ‘We don’t have the drug today.’ This isn’t policy-it’s daily trauma. We’ve got the tools to fix this: domestic API production, stockpiles, mandatory reporting. We just need the will. And maybe a little humility. We’ve been ignoring this for 15 years. It’s not too late to start listening.
And to the people saying ‘it’s just generics’-try being the parent whose kid needs those generics to survive. Then come back and tell me it’s not urgent.
Joseph Cooksey
February 6, 2026 AT 15:11Let’s be real-this whole situation is a masterclass in American dysfunction. We outsource our life-saving chemistry to factories in Gujarat and Wuhan, then wonder why things fall apart. We’ve got a Congress that can’t even pass a budget but somehow finds time to debate whether a senator’s phone records are ‘classified.’ Meanwhile, the FDA’s got 3 interns running a system that tracks 287 life-or-death shortages. The bill’s a Band-Aid on a hemorrhaging artery. What we need is a national pharmaceutical industrial policy. Build domestic manufacturing. Subsidize redundancy. Force vertical integration. Or keep pretending that a spreadsheet update is going to save a cancer patient’s life. Your call.
Justin Fauth
February 8, 2026 AT 11:08China’s making our medicine. Russia’s got the rare earths. And we’re sitting here with a shutdown while our kids go without insulin? This isn’t politics-it’s treason. We let foreign entities control our healthcare supply chain and now we’re surprised when it breaks? Wake up. This isn’t about bills-it’s about sovereignty. We need to nationalize the production of critical drugs. Build the plants. Hire the workers. Stop begging for scraps from foreign governments that don’t care if we live or die.
Lorena Druetta
February 9, 2026 AT 15:54As a healthcare administrator, I must emphasize: the human cost of these shortages is immeasurable. Patients delay treatments. Families deplete savings. Clinics ration care. The proposed legislation, while imperfect, represents a necessary step toward accountability. We must prioritize the integrity of our public health infrastructure above partisan gridlock. The FDA must be fully funded. The reporting mechanism must be operational. And the voices of those affected must be centered-not sidelined.
Zachary French
February 10, 2026 AT 10:38Ohhhhhhhh, here we go again. Another bill. Another ‘solution.’ S.2665? H.R.1160? Sounds like a Microsoft Excel macro. Meanwhile, I called my local pharmacy yesterday. They said they’ve been out of levothyroxine since November. The FDA website? Still says ‘no shortages reported.’ LOL. That’s like saying ‘no fires reported’ while the whole block is burning. And H.R.1160? You mean the bill that doesn’t exist? Yeah, I’ve seen it too. It’s in the ‘pending’ folder with ‘tax reform’ and ‘climate change.’
Here’s the real problem: we don’t need more laws. We need enforcement. We need someone to go to the CEO of a drug company and say, ‘You have 72 hours to fix this or we seize your factory.’ Not a form. Not a report. A SWAT team with a subpoena. But no-instead, we have hearings. And PowerPoint slides. And ‘stakeholder engagement.’ You know what that means? It means we’re all just waiting for someone else to fix it.
Daz Leonheart
February 12, 2026 AT 02:52My sister’s a nurse in rural Kansas. She told me last week they had to use a 1980s-era version of a blood thinner because the new one was out. She said patients didn’t even know the difference. But she knew. And she felt guilty. This isn’t about politics. It’s about showing up. The bill’s not perfect, but if we don’t start somewhere, we’ll keep losing people. Talk to your rep. Call the FDA hotline. Even if it’s just one voice-it matters.
Keith Harris
February 13, 2026 AT 16:21Oh wow, so the solution to a government shutdown is… MORE legislation? You’re kidding me. The FDA’s not working because Congress can’t stop fighting over who gets to eat the last slice of pizza. This bill? It’s a distraction. A feel-good headline for people who think typing ‘#FixTheShortages’ into Twitter fixes anything. We don’t need more rules-we need to fire the people who let this happen. And then we need to rebuild the damn system from scratch. Not with committees. Not with reports. With people who actually know how to make medicine, not just write about it.