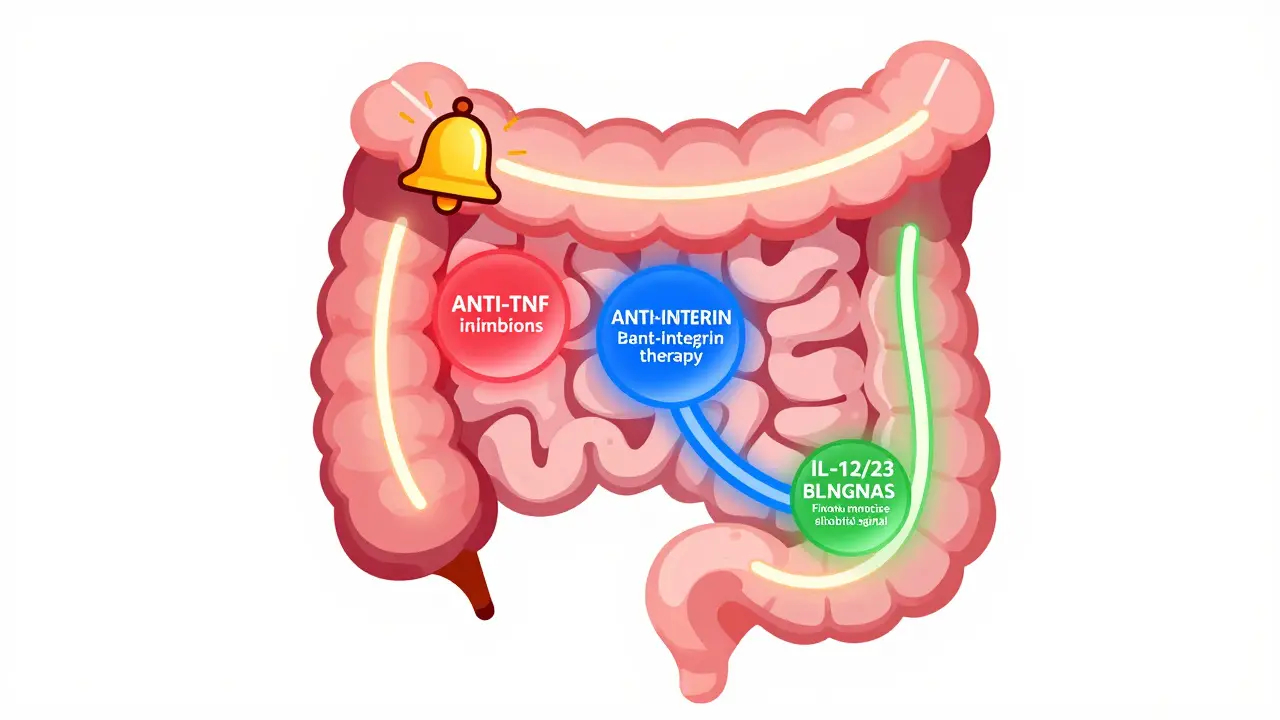
When you're living with Crohn's disease or ulcerative colitis, finding the right treatment isn't just about taking a pill-it's about changing your whole life. For many people, conventional drugs like steroids or immunomodulators stop working. That’s when biologics come in. These aren’t your everyday medications. They’re precision tools designed to target specific parts of your immune system that are causing damage in your gut. Today, three main classes of biologics are used: anti-TNF, anti-integrin, and IL-12/23 inhibitors. Each works differently, has its own pros and cons, and fits certain patients better than others.
What Are Anti-TNF Inhibitors?
Anti-TNF drugs were the first biologics approved for inflammatory bowel disease (IBD). They block tumor necrosis factor-alpha (TNF-α), a protein that drives inflammation in the gut. Think of TNF as a loud alarm bell ringing nonstop in your intestines. These drugs quiet that alarm.
The most common anti-TNF agents are infliximab (Remicade), adalimumab (Humira), golimumab (Simponi), and certolizumab pegol (Cimzia). Infliximab is given as an IV infusion-usually at a clinic-over a few hours. You get doses at weeks 0, 2, and 6, then every 8 weeks after that. Adalimumab is a self-injected shot you can give yourself every other week. Both have been around for over a decade and have helped millions.
But they’re not perfect. About 40% of patients report side effects like infusion reactions (for infliximab) or injection site pain (for adalimumab). More seriously, anti-TNFs increase the risk of serious infections like tuberculosis or pneumonia. The FDA requires special monitoring programs for these drugs because of this. Some people also develop antibodies that make the drug stop working over time. That’s called loss of response. It happens in 6-25% of users.
Still, evidence shows they work well. A 2022 meta-analysis found infliximab had the highest rate of clinical remission in patients who’d never taken a biologic before-better than adalimumab or vedolizumab. In ulcerative colitis, infliximab was ranked as the most effective for inducing remission, even ahead of other biologics. But here’s the catch: while infliximab might be more effective, adalimumab is easier to use. For many, convenience wins.
Anti-Integrin Therapy: The Gut-Selective Option
If you’ve ever heard someone say, “I want a biologic that only works in my gut,” they’re probably talking about vedolizumab (Entyvio). This drug blocks integrins-molecules that help immune cells stick to the gut lining and cause inflammation. Unlike anti-TNFs, which affect the whole body, vedolizumab stays mostly in the intestines. That means fewer systemic side effects.
It’s given as an IV infusion every 8 weeks after three starter doses. Many patients prefer it because they don’t have to worry about lung or nervous system risks. That’s why it’s often chosen for people with a history of multiple sclerosis, latent TB, or other conditions where systemic immune suppression is dangerous.
But it’s slower. While anti-TNFs can start working in 2-4 weeks, vedolizumab often takes 6-10 weeks to show results. One Reddit user wrote, “Switched from Humira to Entyvio... had to wait 10 weeks for full effect, which was brutal.” That delay can be hard for someone in active flare.
On the plus side, vedolizumab has one of the best safety profiles. In patient reviews, 72% reported effectiveness, and only 18% had side effects. It also lowers hospitalization risk better than anti-TNFs in some studies. A 2023 study found patients on vedolizumab stayed out of the hospital longer than those on TNF inhibitors. That’s huge for quality of life.
IL-12/23 and IL-23 Inhibitors: The New Kids on the Block
Ustekinumab (Stelara) was the first in this class, approved for Crohn’s in 2016 and ulcerative colitis in 2019. It blocks interleukin-12 and interleukin-23-two signaling proteins involved in chronic inflammation. Later, newer drugs like risankizumab (Skyrizi) and mirikizumab (Omvoh) came along, targeting only IL-23. This makes them even more precise.
Risankizumab got FDA approval for ulcerative colitis in June 2024. That’s a big deal-it’s now approved for both Crohn’s and UC. In the ADVENT trial, nearly 30% of patients were in remission after a year, compared to just 10% on placebo. That’s strong. And because it’s given as a simple shot under the skin (every 8 weeks after two starter doses), it’s easier than infusions.
These drugs have fewer infection risks than anti-TNFs. No black box warnings for TB or cancer. No need for special monitoring programs. That’s why experts are excited. Dr. Gil Kaplan of the American Gastroenterological Association says IL-23 inhibitors represent the future of IBD treatment.
Ustekinumab is already widely used. It’s given as a 45mg or 90mg shot depending on your weight. Many patients like it because it’s convenient and works well for those who didn’t respond to TNF inhibitors. In fact, it’s often the next step after anti-TNFs fail.
How Do They Compare? Real-World Data
Here’s what the numbers tell us:
| Biologic | Class | Administration | Time to Effect | Common Side Effects | Key Advantages |
|---|---|---|---|---|---|
| Infliximab (Remicade) | Anti-TNF | IV infusion every 8 weeks | 2-4 weeks | Infusion reactions, infection risk | Strongest evidence for remission; fastest onset |
| Adalimumab (Humira) | Anti-TNF | Subcutaneous injection every 2 weeks | 2-4 weeks | Injection site pain, infection risk | Self-administered; widely covered by insurance |
| Vedolizumab (Entyvio) | Anti-integrin | IV infusion every 8 weeks | 6-10 weeks | Mild fatigue, headache | Gut-selective; low systemic risk |
| Ustekinumab (Stelara) | IL-12/23 | Subcutaneous injection every 8-12 weeks | 4-8 weeks | Upper respiratory infections | Good for TNF failures; no TB screening needed |
| Risankizumab (Skyrizi) | IL-23 | Subcutaneous injection every 8 weeks | 4-6 weeks | Mild infections, headache | First IL-23 inhibitor approved for both CD and UC |
What does this mean for you? If you want fast results and can handle clinic visits, infliximab might be best. If you hate needles and IVs, adalimumab gives you control. If you’ve had infections before or have other autoimmune conditions, vedolizumab or an IL-23 inhibitor could be safer.
Cost, Access, and Real-Life Challenges
These drugs are expensive. A single dose of vedolizumab can cost $5,500. Ustekinumab? Around $7,200. Even with insurance, copays can hit $1,000 a month. That’s why manufacturer assistance programs matter. Janssen’s Janssen CarePath helps 95% of eligible patients pay $0-$5 per infusion. AbbVie and Takeda offer similar support.
But not everyone qualifies. A 2023 survey found 25% of IBD patients struggled to get insurance approval for biologics. Some delay treatment because of this. Others switch to cheaper biosimilars-like Inflectra or Cyltezo-which are nearly identical to the originals but cost 15-30% less.
Time is another hurdle. Infusion visits take 3-5 hours. You need to take time off work, arrange transportation, and sit through premedication and monitoring. One patient said, “The 8-hour round trip every 8 weeks is unsustainable.” That’s why many switch to injectables once they can.
And then there’s the emotional toll. Fear of side effects. Worrying about long-term risks. The stress of managing a chronic condition. One patient told me, “I’m grateful it works-but I still lie awake wondering if this is going to kill me.”
What’s Next? The Future of IBD Treatment
The pipeline is full. Drugs like etrolizumab (targeting a different integrin) are in phase 3 trials. More IL-23 inhibitors are coming. Researchers are also testing combinations-like pairing biologics with oral drugs-to get better results.
But the biggest shift isn’t just new drugs-it’s personalization. Doctors are starting to use blood tests and stool markers to predict who will respond to which biologic. Some patients have genetic markers that make them more likely to respond to ustekinumab. Others have high levels of a protein called calprotectin that suggests vedolizumab will work best.
By 2026, we’ll have head-to-head trials like RHEA and VEGA that directly compare infliximab to risankizumab. That’s the next step. Right now, we’re making educated guesses. In a few years, we’ll know for sure.
What Should You Do?
There’s no one-size-fits-all answer. Your choice depends on:
- How severe your disease is
- Your other health conditions
- How much time you can spare
- What your insurance covers
- Your tolerance for side effects
Start by talking to your gastroenterologist. Ask: “What’s the best option for me, not just the most popular one?” Bring up cost. Ask about biosimilars. Ask about patient assistance programs. Don’t be afraid to push for a second opinion.
And remember: biologics aren’t magic. They don’t cure IBD. But for many, they turn a life of pain, hospital visits, and constant worry into something manageable. One woman told me, “Before this, I couldn’t leave the house. Now I’m back at my daughter’s soccer games. That’s worth the shot.”
What’s the difference between anti-TNF and IL-23 inhibitors?
Anti-TNF drugs block a broad inflammatory signal (TNF-alpha) that affects the whole body, increasing infection risk. IL-23 inhibitors target a more specific pathway (interleukin-23) that’s mainly involved in gut inflammation, making them safer and more targeted. IL-23 inhibitors like risankizumab also work better for patients who didn’t respond to anti-TNFs.
Can I switch from one biologic to another?
Yes. Many patients switch due to loss of response, side effects, or cost. Switching from an anti-TNF to vedolizumab or ustekinumab is common and often successful. Studies show about 50-60% of patients respond to a second biologic. But switching within the same class (like from adalimumab to infliximab) is less likely to work.
Why do some people need IV infusions instead of shots?
It depends on the drug’s design. Infliximab and vedolizumab are large molecules that can’t be absorbed through the skin, so they must be given intravenously. Adalimumab and ustekinumab are smaller and can be injected under the skin. IV infusions also allow doctors to monitor for reactions in real time.
Are biosimilars as good as the originals?
Yes. Biosimilars like Inflectra and Cyltezo are nearly identical to Remicade and Humira in structure, safety, and effectiveness. They’re approved by the FDA based on strict testing. Many patients switch without any change in symptoms. The main difference is cost-biosimilars are 15-30% cheaper.
What should I do before starting a biologic?
Get all vaccines up to date-including flu, pneumonia, shingles, and hepatitis B. Avoid live vaccines after starting. Get tested for TB and hepatitis B. Tell your doctor about any history of cancer, heart failure, or nervous system diseases. Some biologics aren’t safe if you’ve had these.
How long do I need to stay on a biologic?
Most patients stay on biologics long-term. Stopping often leads to flare-ups. Some people can taper off after years of remission, but that’s rare and only done under close supervision. Think of it like managing high blood pressure-you need to keep taking it to stay healthy.
If you’re on a biologic, you’re not alone. Over 3 million Americans use them. The goal isn’t just to feel better-it’s to live fully. Whether you’re getting a shot at home or sitting through an infusion, every dose is a step toward reclaiming your life.



Andrew Poulin
March 6, 2026 AT 15:29Susan Purney Mark
March 8, 2026 AT 12:02Bridget Verwey
March 8, 2026 AT 21:22William Minks
March 9, 2026 AT 00:13Adebayo Muhammad
March 9, 2026 AT 18:44Jeff Mirisola
March 10, 2026 AT 04:10Ferdinand Aton
March 10, 2026 AT 04:19Ian Kiplagat
March 11, 2026 AT 07:09Amina Aminkhuslen
March 13, 2026 AT 01:12Pranay Roy
March 15, 2026 AT 00:41Tim Hnatko
March 16, 2026 AT 15:24phyllis bourassa
March 17, 2026 AT 05:52amber carrillo
March 17, 2026 AT 23:44Joe Prism
March 19, 2026 AT 19:08Aaron Pace
March 20, 2026 AT 09:30